Background
Methotrexate is a drug which is often unintentionally involved in medication misadventure.
Because of its unusual dosage requirements (e.g. weekly rather than daily dosing) it is particularly associated inadvertent mistakes by patients and medical staff. This may occur as a result of confusion by patients with other ‘daily’ medications or prescriptions being misinterpreted by nursing staff and pharmacists.
This week’s news has highlighted the high-degree of risk with this medication…

ABC News – 6th June 2016

Mechanism of Action
Methotrexate (which is similar in structure to Folic Acid) acts by competitive inhibition of the enzymes dihydrofolate reductase and thymidylate synthetase. The effect on enzymes associated with immunity leads to a decrease in DNA and RNA synthesis that effects normal cell division.
This mechanism of action explains the features of hair loss and bone marrow suppression normally associated with the drug when given as a therapy for arthritis, psoriasis or as chemotherapy.

Acute overdose
Toxicity does NOT typically occur after deliberate self-poisoning by single ingestion of tablets.
In kids, deaths from single tablet ingestion (one pill can kill) have not been reported but accidental ingestion warrants a hospital visit.
Toxic effects occur at the following levels of ingestion (T0x Handbook):
- Single dose <500mg (5mg/kg in kids) – toxicity unlikely
- Single dose >500mg (5mg/kg in kids) – toxicity possible

How can this happen?
Accidental overdose over a period of days to weeks can lead to lethal bone marrow suppression and can occur if the medicine is taken above the normal dosing for just a few days.
Given that we normally prescribe the medicine weekly, daily dosing can lead to major problems.
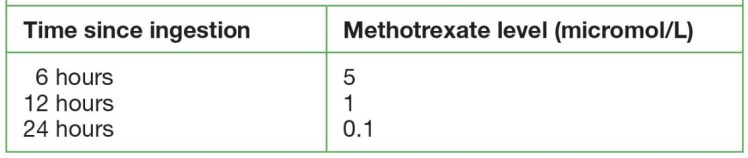
In terms of blood level toxicity the following would raise a concern:

Which patients are at a higher risk of toxicity?
- Patients with:
- Renal disease
- Liver failure
- Other chronic diseases
- Undernourishment
- Extremes of age

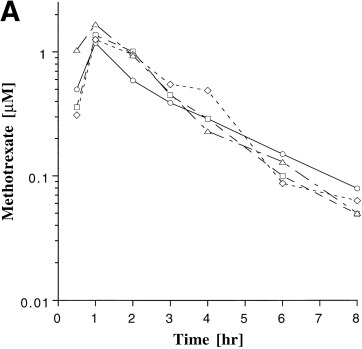
What are the pharmacokinetics?
- After taking the drug peak levels occur within a couple of hours
- The half-life can increase significantly in overdose and renal failure
- The drug is over 80 percent excreted by the kidney unchanged
- (As a result you could consider ‘urinary alkalinisation’ with bicarbonate to increase excretion as a therapy but this is controversial and not routinely recommended)
- Hepatic metabolism produces a nephrotoxic metabolite

What happens to the patient in overdose and what are the signs of toxicity?
- Most patients are not symptomatic after taking a single ingestion of pills
- Features of above therapeutic ingestion include:
- Gastrointestinal (Oral Ulceration, Nausea, Diarrhoea)
- Bone marrow (WCC, HB, PLT)
- this reaches a peak, like other chemotherapy agents after around 10 days
- Hepatic (Liver Injury)
- Renal (Acute Kidney Injury)
- Rare – Severe Skin Problems (Stevens Johnson Syndrome)

What are the treatment options?
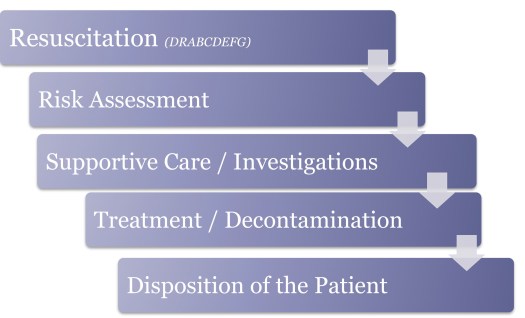
As always in Emergency Medicine we should systematically divide our management plan into phases (e.g. Resuscitation, Specific, Supportive and Disposition).
In Toxicology we amend this approach slightly to include risk assessment:
Resuscitation Phase
- Assessment and Treatment of the ‘ABCs’ are essential (as with any emergency patient).
- Initial tests should include ECG, BSL and paracetamol levels.
Specific Treatment
- With acute overdose consider using charcoal (seek expert help)
- Folinic Acid is considered a ‘specific treatment’:
- Folinic acid – Dose is 15 mg oral/IM/IV
- (End point – aim for Methotrexate Level of <0.05micromol/L)
- Using this depends on risk assessment, drug levels and clinical features.
-
“Folinic acid is indicated if a drug levels cannot be obtained within 24 hours, the patient is symptomatic, renal function is abnormal or drug levels are above the threshold for toxicity.”
- New Treatments:
- Glucarpidase has recently been approved for the treatment of methotrexate toxicity
Supportive Care
- IV fluids are an important part of the overall care of the patient
- (End Point – monitor urine output)
- Seek and treat sepsis (the patient may have severe immunosuppression)
- Consider using granulocyte colony stimulation factor (G-CSF) for haematological complications
Disposition and Monitoring
- All patients require consultation with a friendly toxicologist
- Consult ICU and Haematology where required
- Report the incident where appropriate
- Emergency discharge may be appropriate in asymptomatic patients with normal renal function and low serum levels of the drug after seeking expert advice.

References and Further Reading
- Weidmann A et al . Methotrexate Toxicity During Treatment of Chronic Plaque Psoriasis: A Case Report and Review of the Literature. Dermatology and Therapy. 2014;4(2):145-156
- Tuffaha, H et al. Glucarpidase for the treatment of life-threatening methotrexate overdose. Drugs of Today 2012;48(11), 705–711.
- Balit C et al. Acute methotrexate overdose. Clinical Toxicology 2006; 44(4):411–412.
- Lovecchio F, Katz K, Watts D et al. Four-year experience with methotrexate exposures. Journal of Medical Toxicology 2008; 4(3):149–150
- Intra-theccal overdose
- Schmiegelow K. Advances in individual prediction of methotrexate toxicity: a review. Br J Haematol. 2009 Sep;146(5):489-503
- Murray et al – Toxicology Handbook (2e)